
Main Page | Table of Contents | Journals | Subscriptions | Submissions | Monographs | IBR Grants | IBR Conferences | Hadronic Mechanics | Latest Discoveries | About Us | CV of IBR Members | E-Mail Us

An 83 pages memoir on the foundations of hadronic mechanics and chemistry can be printed out from the pdf file
<R. M. Santilli
ISO-, GENO-, HYPER-MECHANICS FOR MATTER, THEIR ISODUALS,
FOR ANTIMATTER, AND THEIR NOVEL APPLICATIONS IN PHYSICS, CHEMISTRY AND BIOLOGY,>
in press at the Journal of Dynamical Systems and Geometric Theories.
Additional technical presentations are available in
************************************
3.1.B. GENERAL OUTLINE
The monograph reports in a language accessible to the general scientific audience systematic research on new energies and fuels conducted at the particle, nuclear, and molecular levels, that I initiated at Harvard University in 1978 under DOE contracts ER-78-S-02-47420.A000, AS02-78ER04742, DE-ACO2-80ER10651. Santilli then continued the research in collaboration with several mathematicians, theoreticians, experimentalists, solid state physicists, chemists and biologists at The Institute for Basic Research (then in Cambridge, Massachusetts) and at various other institutions under DOE contracts DE-ACO2-80ER-10651.A001, and DE-ACO2-80ER10651.A002. Subsequently, the research was continued under financial support by Hadronic Press, Inc., then in Nonantum, Massachusetts. More recently, the research was completed under logistic and financial support by Toups Technology Licensing, a public company in Largo, Florida. Far from being a presentation of vague academic character, and as stated beginning with Part I, the primary objective of the monograph is strictly pragmatical-concrete-industrial. It consists in the use of hadronic mechanics for the identification of the physical laws, geometries, and conditions under which new clean energies and fuels are possible, and the recommendation of concrete and specific embodiments, called ≥Hadronic Reactors≤, for the best possible realization of said laws, geometries, and conditions. For that purpose, the monograph classifies the reactors into three separate types: Hadronic Reactors of Class I, that are solely based on new effects in the interior of hadrons; Hadronic Reactors of Class II, that are based on new effects in the structure of nuclei with possible contributions from Class I; and Hadronic Reactors of Class III, that are based on new effects at the atomic-molecular structures, with possible contributions of Classes I and II. Numerous Hadronic Reactors under Patent Pending are then outlined. The reactors are solely possible under the validity of hadronic mechanics, and, more particularly, under new structure models of hadrons, nuclei, and molecules, permitted by hadronic mechanics and outlined in Part III, IV and V, respectively, jointly with available experimental verifications. As an illustration, the assumption that three quarks are the constituents of the neutron prohibits any possible utilization of the energy released in its decay, trivially, because the quarks must be confined. On the contrary, the assumption that the physical constituents of the neutrons are one proton and one electron as originally conceived by Rutherford (which model is prohibited by quantum mechanics, yet fully admitted by hadronic mechanics), does indeed permits the utilization of the clean energy of Class I contained in the neutron, because the emission of the structural electron can be stimulated. A similar, mutually exclusive situation occurs between the reactors of Classes II and III and the conventional quantum models of nuclear and molecular structures. Toups Technology Licensing had a crucial role in the completion of the research presented in the monograph, by conducting fundamental anomalous measurements outlined in Part V. These measurements permitted the successful construction of the the first Hadronic Reactor of Class III that has resulted to be over-unity of 2.78 according to the independent certification by Motorfuelers, Inc., a Florida testing laboratory, in its first prototype version (see Section II in this web page).
1 -> I*(t, r, v phi, ...) = 1/T(t, r, v, psi, ...),
and reconstruct the mathematical structure of the theory in such a way to admit the generalized unit as the correct left and right unit. Via mere compatibility arguments one then obtains the SantilliĻs iso-Heisenberg equations
i dA/dt =[A, H]* = ATH - HTA
[r, p]* = rTp - pTr = i I*,
and the Mignani-Myung-Santilli iso-Schroedinger equations
HT|> = E|>
pT|> = - iI*D|>
The reason for selecting a generalized unit is that any other representation of nonlinear, nonlocal and nonhamiltonian interactions is known to be NON-invariant under the time evolution of the theory, or having other catastrophic inconsistencies (page 19), thus having no physical value of any type. By comparison, the unit is the fundamental invariant of any theory, thus assuring the invariance of the numerical results, again, when the mathematical elaboration of the theory is properly lifted. The lifting of the unit then allows three new mathematical and related physical formulations, called iso-, geno-, and hyper-theories (page 18), depending on whether the generalized unit is single-valued hermitean, single-valued nonhermitean and multiple-valued nonhermitean. These theories are used for the representation of closed-conservative reversible systems (such as hadrons, nuclei, and molecules), open-nonconservative irreversible systems (such as open particle, nuclear, and chemical reactions), and multivalued irreversible systems (such as biological structures). Hadronic mechanics represents antimatter is via an anti-isomorphic map called isoduality, that is applicable at all levels, beginning at the Newtonian level, and then ending at the particle level, where it is equivalent to charge conjugation. An important discovery of hadronic mechanics at the foundations of new clean fuels, and energies is that the deep overlapping of the wavepackets or charge distributions of particles, when in singlet couplings (antiparallel spins), implies an attraction so strong to overcome possible repulsive Coulomb forces, thus resulting on new bonds simply not predictable by quantum mechanics, as experimentally verified for the deep correlation-bonding of the two electrons of the helium, the two electrons of the Cooper pair, the electrons in ball lighting, and other events. A main feature of hadronic mechanics is the representation of the capability of particles with the same charge to form bound states at short distances. In turn, these new bonds have permitted the construction of new structure models of hadrons, nuclei, and molecules outlined in Ref. [1], that are at the foundations of Santilli PlasmaArcFlow Reactors, as well as of the new fuels and energies outlined in this page.
3.1.C. PART I: INSUFFICIENCIES OF QUANTUM MECHANICS
This first part begins with the statement of objectives of the series: the identification of the basic physical laws and related technological realizations of energies and fuels, that: are new, in the sense that they cannot be predicted and treated via quantum mechanics; are clean, in the sense that they do not produce radiations, waste, or exhausts harmful to humans and the environment; and are real, in the sense that they are realizable with current technologies in a way having practical, thus industrial and consumer value. Part I then provides a systematic presentation of the limitations of quantum mechanics in particle physics, nuclear physics, superconductivity, chemistry, biology, gravitation, astrophysics, and cosmology. These limitations are a necessary pre-requisite for the presentation of a generalization (also called covering) of quantum mechanics known as hadronic mechanics, that outlined in the subsequent parts. It is stressed that, by no means, the series seeks ≥violations≤ of quantum mechanics, but merely the identification of novel, generally non-Hamiltonian conditions and effects at short distances under which the theory is inapplicable, because not constructed for these new scopes.
3.1.D. PART II: THE NEW MECHANICS
Part II is devoted to the outline of the covering mechanics used in these studies, hadronic mechanics, originally proposed by Santilli in 1978 jointly with its basic equations and related new mathematics. Hadronic mechanics was subsequently developed by numerous mathematicians, theoreticians, and experimentalists, and has now reached operational maturity for applications in the industry. Part II begins with a review of the catastrophic inconsistencies suffered by all generalized theories with a nonlinear, nonlocal or nonunitary structure, due to their lack of invariance, with consequential need of new mathematics for their invariant representation. Part II then outlines three, progressively generalized, new mathematics called iso-, geno- and hyper-mathematics, that have been constructed for the invariant treatment of nonlinear, nonlocal, and nonunitary theories representing closed-isolated-reversible systems, open-nonconservative-irreversible systems, and multi-valued-irreversible systems, respectively. The fundamental assumption of all these mathematics and mechanics is the generalization of the unity, from its trivial value +1 dating back to biblical times, to a positive-definite, but otherwise arbitrary function, matrix, or integro-differential operator. The entire mathematics and mechanics are then reconstructed to admit such a generalized units at all levels,. with no exception (to avoid the catastrophic inconsistencies indicated earlier). All characteristics, interactions and effects outside quantum mechanics (that is, not representable with a Hamiltonian) are represented with the above generalized unit, that, being the fundamental invariant of the theory, does indeed permit the achievement of an invariant, axiomatically consistent formulation of nonlinear, nonlocal and nonunitary effects. as expected in deep overlapping of the wavepackets of particles. Part II then presents an outline of the nonrelativistic and relativistic, iso-, geno-, and hyper-mechanics, with particular emphasis to: 1) The identity of quantum and hadronic mechanics at the abstract, realization free level; 2) The verification by isomechanics of all conventional quantum laws, such as HeisenbergĻs uncertainties, PauliĻs exclusion principle, causality, etc., only expressed via a broader formalism; and 3) A very simple method for the construction of concrete hadronic models accessible to all, thjat consist of simple nonunitary transforms of quantum models. Part II includes a detailed proof of the direct universality of hadronic mechanics, that consists, on mathematical grounds, of its formulation over the most general known numbers and fields, the iso-, geno-, and hyper-octonions (that is, numbers defined with respect to a generalized Hermitean, nonhermitean and nonhermitean multivalued unit). The physical counterpart of the above direct universality is the capability by hadronic mechanics to admit in an axiomatically consistent and invariant way of all infinitely-possible, nonlinear, nonlocal, and nonunitary theories (universality), directly in the frame of the experimenter (direct universality). Readers should be aware that a technical understanding of this direct universality requires a technical knowledge of the catastrophic inconsistencies of conventional generalized theories and their resolution via the new mathematics. particular emphasis is given in this Part II to the identification of SantilliĻs iso-, geno- and hyper-special relativities, including the directly universal liftings of the underlying Minkowskian geometry, the Lorentz-PoincareĻ-symmetry, and the physical axioms. The fundamental representation is the locally varying character of the speed of light within physical media, as established in our environment (such as the refraction of light). As shown in subsequent parts, the deviations from the speed of light in vacuum, and the invariant representation via SantilliĻs isospecial relativity, are at the foundation of all new clean energies and fuels studied in this monograph, and their realization via Hadronic Reactors. Experimental verifications of hadronic mechanics and related new relativities in particle physics, nuclear physics, chemistry and other fields are presented in Parts III, IV and V, respectively. These studies imply the clear identification beyond credible doubts of ConteĻs plagiarism of the basic concepts, laws, and equations of hadronic mechanics and the plagiarism of SantilliĻs isospecial relativity, without the quotation of the specific papers of their origination, in ConteĻs documented knowledge of their existence.
3.1.E. PART III: STRUCTURE OF THE NEUTRON AND NEW ENERGIES OF CLASS I
Part III initiates the application of hadronic mechanics to new energies, beginning with the classification of the new energies into: Class I, when of particle origin; Class II, when of nuclear origin, with possible contributions of Class I; and Class III, when of atomic-molecular origin, with possible contributions of Classes I and II. As a necessary pre-requisite for the study of the new energies of Class I, this Part III outlines the rather numerous experimental verifications of hadronic mechanics in particle physics, astrophysics, gravitation, and cosmology. Part III then outlines new structure models of unstable hadrons with physical constituents emitted in the spontaneous decays with the lowest mode, which models are strictly prohibited by quantum mechanics, yet readily possible with the covering hadronic mechanics thanks to the new nonlinear, nonlocal and nonunitary effects due to deep overlapping of the wavepackets of the hadronic constituents. The new structure models with ordinary physical constituents are at the foundation of new, clean energies of Class I. Particular attention is devoted to the first achievement by Santilli in 1990 of the representation, via hadronic mechanics, of all characteristics of the neutron as a bound state of one proton and one electron totally immersed in its interior according to RutherfordĻs historical legacy. The plagiarism by E. Conte in 1999 of the main equations and results obtained nine years earlier without quotation of the original derivation (in ConteĻs documented awareness), is briefly indicated. Part III then reviews the first experimental verification of RutherfordĻs conception of the neutron conducted by the late Italian priest-physicist don Borghi et al. A subsequent equivalent verification claimed by Conte et al. is indicated too, although with reservations due to the need of independent verifications following ConteĻs dismissal of the scientific priority by don Borghi et. al, with consequential loss of scientific credibility. Part III then presents an example of new energies of Class I, those based on the hypothesis first submitted by Santilli in 1994 that the neutron can be stimulated to decay, thus releasing the rather large energy of 1.294 MeV per decay. The related experimental verification by Gr. Tsagas et al. is also outlined. The resulting new knowledge is applied to new means for the recycling of highly radioactive nuclear waste, via its stimulated decay. Part III concludes with the identification of the physical laws and conditions under which new energies of Class I are possible, and the proposal of various new experiments.
3.1.F. PART IV: STRUCTURE OF NUCLEI AND NEW ENERGIES OF CLASS II
Part IV is devoted to new energies of Class II, those originating at the nuclear level, and contains: 1) a more technical identification of the rather numerous limitations of quantum mechanics in nuclear physics; 2) evidence of the capability by hadronic mechanics of resolving said limitations; and 3) the achievement by hadronic mechanics of the first known exact-numerical representations of various nuclear experimental data, that are currently represented only in an approximate way; 4) a new structure model of nuclei composed by protons and electrons, that is strictly prohibited by quantum mechanics, yet fully permitted by the consistent treatment of RutherfordĻs conception of the neutron of the preceding Part III; and 5) various additional experimental verifications of the treatment of nuclear physics via the covering hadronic mechanics. It is then shown that the emerging new structure model of nuclei does indeed permit the conception and treatment of new, clean energies of Class II (those occurring for totally ionized atoms), essentially based on stimulated nuclear transmutations without the emission of neutrons of other harmful radiations. The latter study begins with the identification of the physical laws, geometries and conditions of new energies of Class II, as predicted by hadronic mechanics. The study then concludes with the presentation of various Hadronic Reactors, that is, specific and concrete embodiments suggested by hadronic mechanics, either for new energies, or for improving the efficiency of existing energies of nuclear type. Particular attention is provided to the origin of thunder. It is shown that thunder cannot be explained in a numerical-quantitative way via conventional chemical-physical means (e.g., the rapid displacement of an excessively minute volume of air), since its explanation requires the instantaneous availability of very large amount of energy (equivalent to hundred of tons of explosive). Evidence obtained via bubbles of air in amber is recalled, according to which our atmosphere contained only 50% of nitrogen about 100 million years ago, thus establishing the existence in our planet of a process responsible for the increase of nitrogen to about 80%. After the exclusion of volcanic, asteroid, or other origin, lightning emerges as the most plausible process synthesizing nitrogen from carbon plus deuteron (or carbon plus protons and electrons). It is shown that the latter hypothesis does indeed permit a numerical;-quantitative explanation of thunder, because of the capability of releasing all the needed energy with a mere efficiency of one stimulated synthesis of nitrogen per ten billion molecules of air. The most important hadronic reactors of Class II are suggested via simple embodiments essentially inspired by lighting, that reproduce it within a liquid rich in deuteron and carbon, or within suitable gases. It is stressed that the stimulated nuclear transmutations here considered occur at about 5,000 C, thus being new, in the sense of have no connection to both, hot and cold fusions. Therefore, hadronic mechanics has permitted the identification of a new class of nuclear reactions with new physical laws, and a probability of practical realization much greater than that of both, hot and cold fusions. Other Hadronic Reactors are based on other processes all verifying the hadronic laws of Class II.
3.1.G: PART V: STRUCTURE OF MOLECULES, NEW, CLEAN, FUELS,
AND NEW ENERGIES OF CLASS III
The study of new energies and fuels of Class V (those occurring at the atomic-molecular level with possible contributions of nuclear and particle type) requires the entire knowledge of the preceding four parts, plus new knowledge on molecular structure presented in this part. Part V begins with an outline of the rather serious environmental problems created by fossil fuels, that are only partially known to the public for evident political reasons, such as: 1) Largest production in our environment of carcinogenic substances via the combustion exhausts, at whose comparison the carcinogenic substances we ingest in food is quantitatively ignorable; 2) Oxygen depletion in our atmosphere at times below the minimum level required for proper heart function; 3) Green house effect due to excessive emission of carbon dioxide over the amount that can be processed by the depleting forests, and other serious problems. Environmentalists are suggested to turn their efforts into scientific activities, that can only occur via the actual measurement of carcinogenic substances in our urban atmosphere, the actual measurement of local oxygen content, and the actual measurement of local carbon-dioxide. These measurements should then be compared with known standards, and the information should then be propagated to the public. The above unreassuring scenario re-affirms the need for new clean fuels, that is, fuels not permitted by quantum chemistry by their very definition (Part I). Part V then passes to the outline of: the rather serious insufficiencies of quantum molecular models; a new model of molecules permitted by hadronic covering of quantum chemistry recently constructed by RR. M. Santilli and D. D. Shillady, which resolves said inconsistencies; and achieves the first representation of molecular characteristics exact to the seventh digit (while quantum chemistry misses about to 2%). Part V then reviews the new chemical species called Magnecules,that are composed of ordinary atoms and molecules under a new strong magnetic bond originating in the plane polarization of the orbits of valence electrons. The rather vast experimental verification of magnecules achieved at independent laboratories by Toups Technology Licensing is outlined, jointly with other advances in molecular structure. It is then shown that the new model of molecular structure permitted by hadronic chemistry, and the new chemical species of magnecules, do indeed permit the prediction and quantitative treatment of new clean energies and fuels of Class III. Particular emphasis is dedicated to the discovery by Santilli at Toups Technology Licensing of a new reactor, called PlasmaArcFlow Reactor (Copyrighted, trademark, and Patents Pending) predicted by by hadronic mechanics and chemistry, that permits the production of clean combustible gases, called MagneGas (copyrighted and Patents Pending) resolving the indicated environmental problems of fossil fuels (no appreciable carcinogenic substances in the exhausts, positive oxygen balance, and carbon dioxide down to small percentages that can be controlled via chemical sponges). The new technology is quite easy in its practical realization, and essentially consists in the flow of a certain liquid within a certain electric arc. The prediction that this process is over-unity for at least 1.69 is presented in Section 3.12 of Part V for independent verifications (see the outcome of its certification in the following Section II of this web page). Part then identifies the hadronic laws, geometries and conditions for new energies of Class III, and shows that hadronic mechanics supports in full, although under novel perspectives, most of the currently known new energies of Class III with clear experimental backing, yet current poor efficiency, including: the electromagnetically pinched deuteron energy; the so-called cold fusion; MillsĻ Blacklight; and other new energies. It is shown that all these new energies have not achieved efficiencies of industrial relevance until now because of their lack of verification of hadronic laws of Class III. A number of Hadronic Reactors of Class III are then proposed, including various Hadronic Fusion Reactors, that are based on the most efficient possible realization of all hadronic laws, geometries, and conditions of Class III. Thanks are expressed to several individuals and corporations in three pages of acknowledgments, with particular reference to: the Department of Energy and the Hadronic Press, Inc., for funding the research over a protracted period of time; the Journal of New energy, for publishing all together the series of five articles; and Toups Technology Licensing, for permitting and supporting measurements on fundamental aspects contrary to the predictions of quantum mechanics and chemistry, which measurements have now acquired a truly basic role for new, clean energies and fuels.
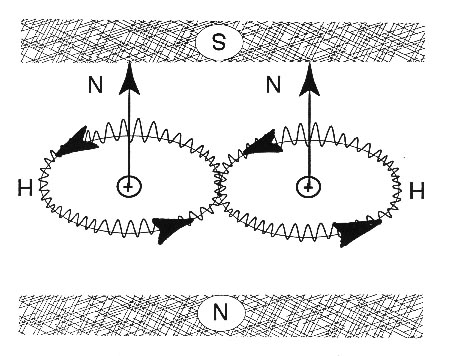
The increased conductivity can be understood by the fact that individual electrons have electric and magnetic fields, while pairs of electrons as in Figure 3.1 have a double charge -2e, yet a virtually null magnetic field. The advantages of the propagation of the latter over the former is then evident, due to the absence of magnetic interactions for the latter and related disturbances in propagation because of interactions with atomic fields. In summary, a current of paired electrons in singlet couplings has an efficiency necessarily bigger than that of a current constituted by individual electrons.
It is well known that quantum mechanics can represent superconductivity only at the statistical level, that is, at the level of an ensemble of large numbers of Cooper pairs, and cannot evidently represent the very foundations of superconductivity, the bonding of electrons in singlet coupling, thus mandating the construction of a covering theory for real scientific advances in the field.
A. O. E. Animalu [3] and, subsequently, Animalu and Santilli [4] have constructed hadronic superconductivity, that is, the generalization of superconductivity permitted by hadronic mechanics. Its main feature is the exact-numerical representation of the bonding of electrons in the Cooper pair as in Figure 3.1, and then the lifting of the conventional theory into a form admitting of nonlinear, nonlocal, and nonhamiltonian interactions due to deep wave-overlappings.
As it is the case for all hadronic theories, hadronic superconductivity coincides with conventional superconductivity for mutual distances sufficient to render ignorable nonlinear, nonlocal, and nonhamiltonian effects (or render ignorable wave-overlappings), and introduces new nonunitary effects only at short distances. In this sense, hadronic superconductivity is indeed a real covering of quantum superconductivity. Hadronic superconductivity is also divided into iso-, geno-, and hyper-conductivity for the representation of closed-reversible, open-irreversible, and multi-valued irreversible events, respectively.
Besides achieving an exact-numerical representation of experimental data, the most important function of Animalu-Santilli isosuperconductivity is that of identifying the necessary conditions under which the superconducting temperature can be increased. These conditions are evidently linked to technological means for the production of Cooper pairs in sufficiently high numbers to reach practically appreciable results. In turn, these means are deeply linked to molecular models, as indicated in the next section.
As indicated in Sect. 2.7, the over-unity of Santilli's PlasmaArcFlow Reactors is crucially dependent on the electric arc within liquids to be mostly constituted by a current of electron pairs, rather than individual electrons, thus being superconducting. For details, readers are suggested to consult the original references [1,2,3,4]
1) Lack of a sufficiently strong molecular bonding force. The forces currently assumed in molecular bonds (the exchange, van der Waals, and other forces) were conceived for nuclear structures, in which field they are known to be weak. This is the historical reason that demanded the additional introduction in nuclear physics of strong forces. Moreover, the classical average of currently assumed, bonding forces implies an identically null force between different atoms, as anybody can verify. Thus, the current description of molecular structures according to quantum chemistry misses the equivalent of the strong force in nuclear structures.
2) Impossibility to explain why the hydrogen, water, and other molecules have only two hydrogen atoms. The currently used bonding forces of nuclear type were conceived for an arbitrary number of constituents, as an evident necessary condition to have meaning for nuclei. Any graduate student in physics or chemistry can, therefore, prove that, under the current models of molecular, the hydrogen molecule can be H_3 or H_8, or H_27, and the water molecule can be H_3O, H_12O, H_27O, etc.
3) Lack of accurate representation of molecular binding energies and other molecular characteristics without manipulation of basic principles. As well known, a historical amount of about 2% of the experimental values of molecular binding energies is still missing in quantum chemical representations, e.g., in self-consistent treatments.
4) More accurate representations of molecular characteristics imply departures of basic quantum axioms and laws. More accurate representations of molecular binding energies have been recently achieved, but they require structural modification of the Coulomb law with Gaussian and other factors called ''screenings''. However, Gaussian screened Coulomb forces do not allow the existence of the hydrogen atom, imply necessary instabilities of all electron orbits, and prohibit the use of the very notion of ''quantum of energy'', because quanta are emitted or absorbed only between stable orbits, as well known. A similar occurrence holds for variational methods that, besides admitting an unlimited number of free parameters of completely unknown physical or chemical origin (thus being of purely mathematical character), can be proved by graduate students to violate the very conditions to preserve a ''quantum'', e.g., because variational solutions cannot be solutions of genuinely quantum Schrodinger equations (otherwise the latter would have permitted exact representations).
5) Impossibility to conduct meaningful thermochemical calculations. The 2% currently missing in the binding energy is misleadingly small, because it implies an error of about 950 Kcal/mole, while an ordinary chemical reaction implies about 20 Kcal/mole. Therefore, quantum chemistry implies thermochemical calculations in which the error can be 50 times bigger than the energy considered, thus preventing and seriously scientific thermodynamical calculations
6) Excessive times in computer calculations. Computer calculations in chemistry are known to require up to months of continuous running time, despite the use of the most modern possible computers. This is a clear indication of the insufficiency of the theory, rather than of the computer.
7) Major disagreement between the correlations used in current orbital theories and experimental evidence. Current orbital theories use an arbitrary number of electrons in their correlations, while experimental evidence establishes that correlation only occurs for electron pairs. This evidence establishes an additional insufficiency of the theory (see [1] for technical details).
8) Lack of accurate representations of molecular electric and magnetic moments. In fact, current representations are sometimes wrong even in the sign, let alone the value.
9) Prediction by quantum chemistry that all molecules are ferromagnetic (Figures 3.3), that is in evident, dramatic disagreement with experimental evidence.
Under so many fundamental insufficiencies and sheer inconsistencies, real scientific studies of ''new'' fuels and energies based on quantum chemistry are unwarranted, thus justifying the use of covering theories.
3.3.B. HADRONIC CHEMISTRY
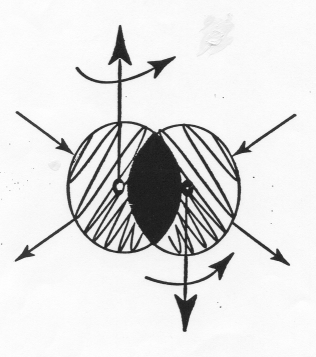
It is evident that the origin of the insufficiencies and inconsistencies of quantum chemistry outlined above are not due to quantum chemistry itself, but rather to the underlying theory, quantum mechanics. It is today known that the indicated problems are due to the absence of a meaningful representation by quantum mechanics of the conditions of deep overlappings of the extended wavepackets of electrons, precisely as occurring in valence electron bonds (Figure 3.1).
The insufficiencies and inconsistencies of quantum chemistry in molecular structures establish that the discipline that is exactly valid for the structure of the individual hydrogen atom (quantum mechanics), cannot possibly be exact for the bonding of two hydrogen atoms into the hydrogen molecule. This is due to the appearance in the H-H structure of short-range, nonlinear, nonlocal, and non-Hamiltonian effects as depicted in Figure 3.1, that are completely absent in the individual H structure, thus confirming the need in chemistry for a more adequate theory.
Thanks to the preceding achievement of mathematical, theoretical, and experimental maturity by hadronic mechanics and superconductivity, R. M. Santilli and D. D. Shillady [5,6,7] have constructed a generalization-covering of quantum chemistry under the name of hadronic chemistry, that is also based on the novel iso-, geno-, and hyper-mathematics (page 18). Therefore, hadronic chemistry contains three corresponding branches also called iso-, geno-, and hyper-chemistry, that are used for a more accurate representation of conservative-reversible molecular structures, irreversible chemical reactions, and multi-valued biological systems, respectively.
Since this page is devoted to molecular structures, that are notorious conservative and invariant under time reversal, we shall solely use isochemistry. Unless otherwise specified, the terms ''hadronic chemistry'' are referred in this page to its isotopic branch.
In essence, isochemistry has permitted the construction of a new model of molecular structure, known under the name of Santilli-Shillady isochemical model of molecular bonds. Its main feature is the assumption that pairs of valence electrons from two different atoms couple themselves in a singlet quasi-particle state at short distances, called isoelectronium, with main characteristics
Charge: 0; spin 0; magnetic moment 0; rest energy 1.0 MeV (max);
Radius 6.8432329 x 10^-11 cm = 0.015424288 bohrs
that orbits around both nuclei in an oo-shaped orbit around the respective nuclei. Such a molecular model is similar to that of planets in certain binary stars.
It should be indicated that, as it is the case for the electric pairs of the helium, and the Cooper pair in superconductivity, the Santilli-Shillady isoelectronium is not a permanently stable particle, due to exchange and other effects.
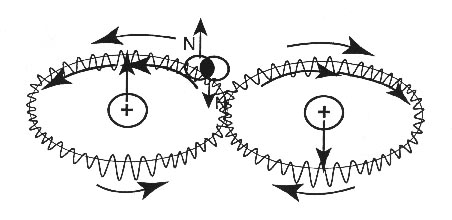
FIGURE 3.4: A schematic view of Santilli-Shillady isochemical
model of the Hydrogen molecules [5] with the two electrons bonded in singlet coupling into
the isoelectronium, that describes an oo-shaped orbit around the two nuclei. Note the
opposite direction of rotation of the isoelectronium in the two o-branches, with
consequential opposite magnetic moments. This configuration prevents the acquisition by the hydrogen
molecule of a net magnetic polarity, thus avoiding the inconsistency of quantum chemistry of Fig. 3.3. The model is
evidently considered at absolute zero degrees K and in the absence of all rotational,
vibrational and other motions. Note also that isoelectronium cannot be permanently stable [5,6,7].
A feature of the isoelectronium of particular significance for new energies is that of having an essentially null intrinsic magnetic moment (in first nonrelativistic approximation, with non-null quadruple and higher moments). The above new conception of molecular structure has permitted the resolution in Refs. [5,6,7] of all insufficiencies and inconsistencies of quantum chemistry outlined above. In fact:
1) Isochemistry introduces for the first time a new strong, attractive force between two valence electrons suitable to represent the strength of molecular bonds, In different terms, the new hadronic force between valence electrons is the equivalent, missing in quantum chemistry, of the strong force in nuclear structures.
2) Isochemistry explains for the first time why the hydrogen, water and other molecules have only two hydrogen atoms. Once two valence electrons are bonded into the singlet isoelectronium, they become a Boson and, as such, reject the bonding of any additional electron (that is a Fermion).
3) Isochemistry has permitted the first representation of molecular binding energies that is accurate to the seventh digit.
4) The above accurate representations occur under the exact validity of the basic axioms of isochemistry without ad hoc adulterations.
5) Since the representation of binding energies is accurate to the seventh digit, isochemistry permits, for the first time, accurate thermochemical calculations.
6) Isochemistry reduces computer usage at least 1,000 times. This is achieves via power series that converge much faster than those of quantum chemistry.
7) Isochemistry allows correlations solely among pairs of electrons at all levels of study.
8) Isochemistry has provided representations of electric and magnetic moments also accurate to several digits [and having the correct sign].
9) Isochemistry has resolved the inconsistent prediction by quantum chemistry that all molecules are ferromagnetic [Figures 1.8 and 1.9].
3.3.C. SANTILLI'S NEW CHEMICAL SPECIES OF MAGNECULES
In this section we shall outline Santilli's discovery of Magnecules^TM made in 1998, by following mostly ad litteram the presentation of their original publication, Ref. [8].
The way according to which atoms can combine to form the various substances of our sensory perception, was scientifically established in the last century, thanks to the work by Avogadro (1811), Canizzaro (1858), and several others, following the achievement of scientific measurements of atomic and molecular weights. These studies produced the contemporary notion of molecules and of its underlying valence.
Until 1997, molecules remained the sole known way according to which atoms can combine to form ordinary substances. The notion of valence has also remained the dominant mechanism for the bonding of atoms, although, with the passing of the decades, it was specialized into covalence, ionic valence, metallic valence, and others (see any quantum chemistry book).
In 1998 R. M. Santilli [8] discovered basically new means for atoms and molecules to bond themselves into clusters that are stable at ordinary conditions. Santilli called the new species magnecules to denote the magnetic nature of their origination and bond, and to distinguish them from molecules holding for conventional valance bonds.
Thanks to an invaluable backing by Toups Technology Licensing, the notion of magnecules received, also in 1998, numerous experimental verifications at independent analytic laboratories, that are outlined in Sections 4 and 5. In this process, a new technology was established for the creation of magnecules that is today known as the TTL technology of magnetically polarized gases.
The sole force fields in the atomic structure studied by chemists prior to Refs. [8,9] have been the electric field of the individual peripheral electrons, and the intrinsic magnetic field of nuclei and electrons. It was proved a century ago that these fields can only produce valence bonds, thus explaining the reason why the notion of molecules was the only one admitted for about two centuries.
Santilli's [loc. cit.] main discovery has been the identification of a a new force field in the atomic and molecular structures, thus permitting a new chemical species.
Since the inception of atomic physics, peripheral electrons have been assumed to have a spherical distribution around nuclei, that is indeed the case under ordinary conditions, as experimentally established. However, electrons are charged particles, and all charges rotating in a plane orbit create a magnetic field in the direction perpendicular to the orbital plane, generally assumed with the North polarity in the semi-space seeing a counter-clockwise rotation. Moreover, it is known that the distribution in space of electron orbits can be controlled via the use of external magnetic fields.
Santilli's [loc. cit.] main hypothesis is that the spherical distribution at least of the valence electrons in the natural state of atoms can be controlled via suitable external magnetic fields, and, in first approximation at absolute zero degree, can be polarized a plane (the actual distribution being toroidal). In turn, the transition of the electrons orbits from a spherical; distribution to a plane polarization creates a basically novel magnetic field that does not exist in the natural state of the atoms.
Moreover, Santilli [loc. cit.] computed the magnetic field created by the polarized orbiting electron of the hydrogen atom, and showed that its numerical value is 658.50 times bigger than that of the nucleus (the proton), while the magnetic moment of the polarized isoelectronium orbit in the hydrogen molecule is 1,316.33 times bigger than the intrinsic magnetic moment of the nucleus. Such a high value of the field was expected from the fact that the orbital speed of electrons approaches the speed of light, thus resulting in very strong magnetic fields associated with plane orbits. These numerical values have been independently verified by Aringazin and Kucherenko [10] and others.
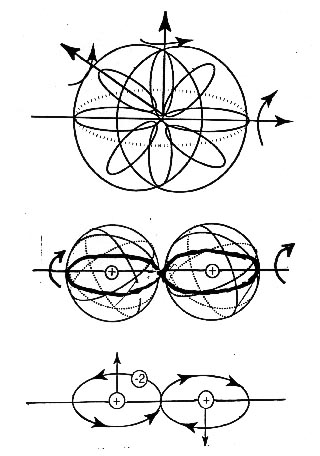
FIGURE 3.5: A schematic view of the new technology of magnetically polarized gases at Toups Technology Corporation, here presented for the simplest possible
case of the hydrogen molecule. The technology essentially consists of electric and/or magnetic and/or
electromagnetic means to eliminate the rotational motion of the orbits of the
valence electron pairs. The upper view shows an H-molecule in its conventional spherical state. The middle view shows the same molecule in which the rotation of the two H-atoms has been eliminated via external magnetic and/or other fields, although each H-atom preserves its spherical distribution. The lower view shows the polarization of the orbit of the valence electrons in a plane (actually within a
toroid). In the last case, we have the emergence of the magnetic moment due to the rotation of the electrons in plane orbits , that is simply absent in conventional conditions of the molecule.
Other possible origin for the bond of the new chemical species must be eliminated due to known insufficiencies. For instance, the intrinsic magnetic moments of nuclei cannot possibly produce a molecular bond, first, because these fields are too weak, and, second, because they are too far from each other on atomic scale. Similarly, the electric polarization of atoms (i.e., their deformation in such a way to offer the dominance of one charge in one side, and the other charge in the opposite side) cannot form a stable molecular bond, because of the known instability of the electric polarization itself.
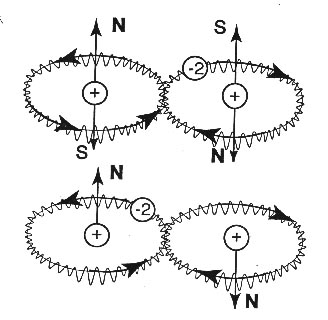
FIGURE 3.6: A schematic view of the simplest possible Santilli's elementary magnecule H2xH2 composed of two plane polarized
hydrogen molecules in mutual magnetic bond. Note that, once the magnecule has been formed,
it is stable (at ordinary conditions) because of the high value of the magnetic forces at
the small inter-atomic distances of the bond. Note also that the elementary magnecule is
considered here at absolute zero degree Kelvin and in the absence of any motion. At
ordinary temperature, the magnecule is predicted to appear as a sphere of
the same size as that of one conventional hydrogen molecule, yet possessing a molecular
weight very close to that of the helium. Similar elementary magnecules are predicted for H2xO2,
O2xO2, COxCO, COxCO2, etc.
The above model of magnecules can be represented as follows. Denote the valence bond according to isochemistry with the symbol " -" and represent the new magnetic bond with the symbol " x ". A conventional, un-polarized hydrogen molecule is then written as H-H. When polarized, the same molecule acquires the structure H{up}-H{down}. By comparison, the same molecule according to quantum chemistry would have the structure H{up}-H{down}{up}that is in disagreement with experimental evidence.
The "elementary magnecule" of the Hydrogen (Fig. 3.6) can then be written
( H{up} - H{down}) x (H{up} - H{down}) ,
where the pairing H{up} x H{up} is the most elementary possible one, that of two hydrogen atoms one against the other in which the South polarity of one atom is bonded to the North polarity of the other atom.
Needless to say, isochemistry predicts the possibility that additional hydrogen molecules or individual H-atoms can bond themselves to the above elementary magnecule, resulting in magnecules of the type
( H{up} - H{down}) x (H{up} - H{down}) x (H{up} - H{down}) x H{up} ,
The above model permits a quantitative understanding of the experimental results presented in Sections 4 and 5. First, the model permits the identification of the magnecules of Fig. 1 (such as peaks with amu 124, 228, 254, 355, 479, etc. that cannot exist for quantum chemistry) as clusters of polarized atoms H{up}, O{up}{down}, C{up}{down} and all their possible molecules also in polarized form, resulting in the generic structure
( H{up} - H{down}) x (O{up}
- O{down}) x [C{up} - O{down}) x
(H{up} - O{down}{up}
- H{down}) x (O{up} - C{down}{up} -O{down}) x H{up}
. . . ,
Quantitative interpretation of Anomalies 1) to 5) then follows and will be studied in detail in a technical paper. Note the crucial role of hadronic mechanics [5] in these results.
A word of caution should be voiced here to point out the complexity of the magnetic
moments of the isoelectronium orbits for all structures more complex than the hydrogen. As
an example, the water molecule has two isoelectronia, one per each H-O dimer, with
symmetry axes intersecting at the known 105į angle, plus the additional six electrons of
the oxygen, resulting in a rather complex magnetic structure whose systematic study is
under way [8].
In summary, we can introduced via the following:
DEFINITION [8,9]: Santilli's electromagnecules, or more simply magnecules, constitute a new chemical species in gases, liquids or solids that is composed of isolated atoms H, C, H, etc., dimers OH, CH, etc. and molecules H2. CO, etc., under a new magnetic bond due to the toroidal polarization of the orbits of [at least] the valence electrons under strong electric and magnetic fields, which species is stable at ordinary conditions of temperature and pressure, and is characterized by the following properties:
3.3.D. CREATION OF MAGNECULES IN GASES, LIQUIDS, AND SOLIDS.
All gases produced via a subnmerged electric arc possess a magnecular structure that is enhanced by the
PlasmaArcFlow^TM process. The outline of magnecules of this web page will be referred hereon to Magnegases^TM with the understanding that all gases of Refs. [11] have Santilli's magnecular structure.
I) The new species has variable molecular weight, depending on the number of atoms and molecules possessing the needed magnetic polarization, which molecular weight can be a multiple of the conventional molecular weight for the same atomic constituents;
II) The new species is identified in mass spectrometry by new peaks that result to be "unknown" in the computer search among all known conventional molecular structures;
III) The new peaks identified in the mass spectrometry have no infrared [for gases] or ultraviolet [for liquids] signature at all, as a necessary condition not to have a valence bond, the sole infrared or ultraviolet signatures being those of the conventional atoms or molecules constituting the new species;
IV) The new species has anomalous physical and chemical characteristics, including anomalous infrared or ultraviolet signatures, anomalous penetrations throught other substances, anomalous adhesion to other substances, anomalous viscosity, etc.;
V) The new chemical species, its peaks, and all its anomalous features disappear at a sufficiently high temperature [the Curie point of the new species] evidently varying from substance to substance.
Magnecules are called: elementary when composed of only two molecules; magneplexes when entirely composed by several identical molecules; and magneclusters when composed by molecules of different type.
It was easy to see that one of the strongest, readily available sources of magnetic fields in gases is an electric arc. Therefore, Santilli predicted that magnecules exist in gases and liquids exposed to an electric arc. In this section we outline separately the experimental evidence for magnecules in gases, liquids, and solids.
An electric arc in a water-base liquid decomposes the various compounds by forming a plasma at about 5,000 C of mostly ionized atoms of hydrogen, oxygen, carbon and other elements, that combine in a variety of ways, forming nonexplosive combustible gases with clean emission exhausts. The new gases cool down in the surrounding liquid, and bubble to the surface where they are collected with various means.
Santilli's main hypothesis [8] is that, at the time of their formation under an electric arc, gases H2, CO, CO2, O2, etc. do not have a conventional structure because the orbits of [at least] their valence electrons are mostly polarized in a plane due to the very intense magnetic field surrounding the electric arc (of the order of 10 Tesla or more). In turn, such a polarization implies the creation of strong magnetic moments, resulting in new magnetic bonds constituting the magnecules.
Conventional mass spectrometric analyses, after adjustments due to possible air contamination (indicated by nitrogen content) and averaging over various methods of production, suggested the following:
Conventional chemical composition of Magnegases:
However, the above chemical analysis is in dramatic disagreement with a variety of experimental evidence. For instance, the heaviest molecule present in Magnegases^TM in macroscopic percentage is CO2 with a molecular weight of 44 amu. However, ,GC-MS measurements have shown the presence of macroscopic peaks all the way to 1,000 amu, that evidently have no conventional explanation via quantum chemistry.
Numerous other discrepancies existed between the predictions of quantum chemistry and the experimental evidence of Magnegases, such as anomalous passage of hydrogen through surfaces, anomalous deflation of balloons, anomalous attraction of floating balloons by metal beams, and other behavior dramatically beyond the prediction of quantum chemistry.
Therefore, Santilli conjectured that all gases produced via an electric discharge are indeed composed by conventional molecules under a special magnetic bond, thus resulting in a new chemical species.The conjecture was confirmed by a variety of GC-MC/IRD measurements reviewed below.
Santilli [8] additionally constructed Magnecules in liquids via methods essentially similar to those for gases. In fact, liquid magnecules were first constructed via the exposure of aromatic oils to strong magnetic fields, and subsequently detected via LC-MSD/UVD. Additional, much more evident liquid magnecules were constructed by passing the liquid through electric arcs, according to the PlasmaArcFlow^TM technology.
Finally, Santilli [loc. cit.] constructed magnecules in solids via the mere freezing of liquids with a magnecular structure.
3.3.E. ANOMALIES OF MAGNECULES
Anomaly 1: Appearance of unexpected heavy MS peaks.
H2: 48 %
CO: 44 %
O2: 5 %
CO2: 2 %
Misc. struct. in ppm: 1 %.
The experimental verification of gas magnecules requires the detection of a number of anomalies that can be summarized as follows:
Gas magnecules are generally heavier than the heaviest molecule in a given gas. Peaks in the GC-MS are therefore expected in macroscopic percentages with molecular weights bigger than the heaviest molecule. These heavy composites should not provide MS peaks according to quantum chemistry, thus constituting an anomaly. As an example, by ignoring heavy compounds in parts per million [ppm], Magnegases^TM should have no large peak in the GC-MS with more than the CO2 molecular weight of 44 a.m.u. The existence of heavier large peaks would establish this first anomaly.
Anomaly 2: "Unknown" character of the unexpected heavy peaks.
To provide the initial premises for magnecules, the peaks of Anomaly 1 should result in "unknown" in the search by the GC-MS computer in its memory banks of conventional molecules, usually including about 150,000 molecules.
Anomaly 3: Lack of IR signature of the "unknown" peaks.
Another necessary condition to have magnecules is that the "unknown" peaks of Anomaly 1 should have no infrared signature at all. According to established evidence, all gases with a valence bond must have a well defined infrared signature [with a few exceptions of spherically symmetric molecules, such as hydrogen]. In the event the peaks of Anomaly 1 do have an infrared signature, they can be constituted by new yet conventional molecules not identified before. The only infrared signatures of any given gas magnecule should be those of the conventional molecules and atoms constituting the cluster itself. As an illustration, the only admissible infrared signatures of magnecule {O2}x{O2} are those of the conventional molecules O-O and C-O.
Anomaly 4: Mutation of conventional IR signatures.
Anomaly 5: Mutation of magnecules.
Anomaly 6: Mutated physical characteristics.
Anomaly 7: Anomalous adhesion.
Anomaly 8: Increased penetration through substances.
Anomaly 9: Increased energy release.
All the above anomalies are expected to disappear at a sufficient high temperature, evidently varying from gas to gas (Curie point), while the anomalies are expected to be enhanced at lower temperature and survive liquefaction.
Liquid magnecules have essentially the same anomalies, while those of solid magnecules are evidently different due to lack of fluidity, thus requiring a special study.
For instance, Anomalies 1 through 6 should be established by GC-MS. However, most GC-MS machines are not suitable to detect magnecules because they usually operate at temperatures generally higher than the Curie point of the magnecule to be detected. In this case, the measuring equipment itself destroys the very features to be measured. The few GC-MS that are suitable for the tests should be operated in a way significantly different than the conventional one, again, to prevent destroying the very features to be measured.
Additional difficulties are created by the rather universal tendency in analytic laboratories to throw a conventional interpretation at any peak in the GC-MS, without considering any IR verification at all. When dealing with magnecules, such conventional "explanations" are vacuous and of generally of non-scientific nature, because the conventional molecular interpretation of peaks in the GC-MS are completely disproved by the lack of IR signatures [as recalled earlier, with very few exceptions for low a.m.u. that do not apply to large magnecules in the 500 a.m.u range, clear IR signatures are a necessary condition to have a conventional valence bond].
The above tendency of throwing conventional molecular interpretations of any GC-MS peak is so widespread, that GC-MS equipment are not generally equipped with an IRD, as it is usual in academic analytic laboratories. In view of the above occurrences, to prevent a waste of time without real science, researchers are strongly recommended to prevent, prohibit, or otherwise avoid any measurement of magnecules with GC-MS equipment without IRD.
Additional difficulties are of human nature, and due to the fact that analysts with long and outstanding experience in conventional analytic procedures, are generally reluctant to change their methods, thus preventing the measurement of new characteristics.
The necessary conditions for scientific measurements of gas magnecules with gas chromatographic, mass spectroscopic equipment are the following:
Condition 1: GC-MS should permit measurements of peaks at ordinary temperature (say, 10 C - 30 C), and the feeding lines should be cryogenically cooled.
Condition 2: GC-MS must be equipped with IRD.
Condition 3: GC-MS/IRD should be equipped with feeding lines of at least 0.5 mm ID.
Condition 4: GC-MS should be set to detect peaks at molecular weights where the analyst usually expects none.
Condition 5: The ramp time should be the longest admitted by the GC-MS/IRD and of at least 25 minutes.
Numerous other conditions exist for GC-MS_IRD to be effective in the detection and identification of magnecules. They are of more technical nature and will be indicated whenever needed.
Besides the problems in identifying an appropriate GC-MS_IRD, additional difficulties exist in the identification of other instruments capable of providing effective measurements of the other Anomalies 7-9, again, because the available instruments have been designed for the measurement of conventional rather than anomalous features.
As an illustration, by far the most difficult measurement of Magnegases^TM has been the achievement of credible scientific values of energy content in British Thermal Units [BTU] per cubic foot [cf]. By comparison, the measurement of the BTU/cf of gases with a conventional chemical structure is so elementary nowadays to be computerized following GC-MS data.
Significantly, all methods currently available to measure the BTU/cf failed to provide results of any credibility for gases with magnecules. This is the case of computerized computation of BTU/cf from GC-MS results] and some conventional calorimeters did not even allow Magnegases^TM to burn, let alone to make a measurement. This is the case of the computerized calorimeters for methane whose air intake is too big for the combustion of all Magnegases^TM and its required adjustment is outside the range of the computer program.
Similar difficulties were encountered via the use of EPA analytic techniques, evidently because they are not designed to detect anomalies.
The conditions for scientific measurements of magnecules in liquid are the same as those for gas magnecules, except for the use of Liquid Chromatography Mass Spectrometers [LC-MS] equipped with UltraViolet Detector [UVD].
3.4.B. EXPERIMENTAL EVIDENCE OF SANTILLI'S MAGNECULES IN GASES
Thanks to a professional cooperation by the NTS analysts, the equipment was set at all the unusual conditions indicated earlier. In particular, the equipment was set for the analytic method VOC IRMS.M utilizing an HP Ultra 2 column 25 m long with a 0.32 mm ID and a film thickness of 0.52 Ķm. I also requested to conduct the analysis from 40 a.m.u. to the instrument limit of 500 a.m.u. This condition was necessary to avoid the CO peak of magnegas at 28 a.m.u. that is so large to dwarf all other peaks.
Moreover, NTS analysts were fully cooperative in implementing unconventional settings of the instrument. In fact, the GC-MS_IRD was set at the lowest possible temperature of 10 C; the biggest possible feeding line with an ID of .5 mm was installed; the feeding line itself was cryogenically cooled; the equipment was set at the longest possible ramp time of 26 minutes; and a linear flow velocity of 50 cm_sec was selected. A number of other technical requirements are available in the complete documentation of the measurements reproduced in App. A of monograph {7b}.
The analysts first secured a documentation of the background of the instrument prior to any injection of the magnetically polarized gas in view of Anomaly 7, that is evidently expected to alter the background due to anomalous residues in the instrument after the completion of the tests.
Following a final control that all requested conditions were in place, NTS analysts Louis A. Dee and Norman Wade injected the polarized gas into the HP GC-MS/IRD and initiated the tests. The results constitute the first direct experimental evidence on the existence of magnecules in gases.
After waiting for the 26 minutes of the ramp time, sixteen heavy anomalous peaks appeared in the MS screen, thus providing direct experimental verification of Anomaly 1 [Figure 3.7].
The polarized gas contains about 5% of CO2 as per various chemical analyses conducted at high temperature, after adjustment due to air contamination. The spectrum of Figure 3.7 is from 40 a.m.u. to 500 a.m.u. Comparative inspection of the various peaks and their size establishes that they represent macroscopic percentages of compounds that, according to quantum chemistry, should not exist in the gas considered, thus providing experimental evidence for Anomaly 1 beyond scientific doubt.
I should report that, at the appearance of the peaks of Figure 3.7, analyst Norman Wade stated "I have not seen something like these peaks in twenty five years of analyses".
The analysts then passed to the identification of the individual peaks of Figure 3.7. As illustrated in Figure 3.8 [see the complete documentation of Ref. [9] for more data], all sixteen MS peaks of Figure 3.7 resulted to be "unknown", following the computer search in the memory banks of the McClellan Air Force Base comprising about 140,000 molecules, thus providing direct experimental verification of Anomaly 2.2.
Note that there is no identifiable CO2 peak in the MS spectrum of Figure 3.7. This absence is, per se, highly anomalous, because the CO2 peak is well identifiable in conventional GC-MS measurements of the same gas at high temperature. In fact, the absence of the CO2 peak in the MS range 40 amu to 500 amu is evidence that it is bonded in the remaining peaks of the spectrum.
After completing and documenting the MS data, the analysts passed to measurements at the IRD. To their surprise, none of the sixteen peaks of Figure 3.7 had any infrared signature at all, thus providing direct experimental verification of Anomaly 3 [Figure 3.9]. The IR scan for the MS peaks of Figure 3.7 only shows a peak belonging to CO2, namely, to one of the components of the peaks of Figure 3.7. The IR signature of the other components do not appear in Figure 3.9 because they are behind the left margin of the scan.
The analysts then established [Figure 3.10] that the IR peak of CO2 is itself anomalous (mutated), thus providing experimental evidence for Anomaly 4.
The infrared signatures of the molecules constituting a magnecules are expected to be mutated, in the sense that the shape of their peaks is not the established one. This is another anomaly of magnecules expected from the polarization of the orbits of the valence and other electrons. In fact, this polarization implies space distributions of the orbitals different than the conventional ones, thus resulting in a deformation of the shape of the IR peaks. Moreover, the same polarizations are expected to create additional strong bonds within a conventional molecule, that are expected to appear as new IR peaks. Still in turn, such an internal mutations of conventional molecules have far reaching scientific and technological implications, as will be shown.
While molecules preserve their structure at conventional temperatures and pressures, this is not the case for
magnecules, that are expected to mutate in time, that is, to change the shape of the MS peaks due to change in their constituents. Since we are referring to gases whose constituents notoriously collide, magnecules can break-down into parts during collisions, which parts can then recombine with other magnecules to form new clusters. Alternatively, magnecules are expected to experience accretion [or emission] of polarized conventional atoms or molecules without necessarily breaking down into parts. It follows that the peaks of Anomaly 1 are not expected to remain the same over a sufficient period of time for the same gas under the same conditions.
Magnetically polarized gases are expected to have mutated physical characteristics because the very notion of polarization of the orbits implies a smaller average molecular volume. Mutations of other physical characteristics are then consequential.
Magnetically polarized gases are expected to have anomalous adhesion to walls of disparate nature as compared to the same unpolarized gas. This is due to the well known property that magnetism can be propagated by induction, according to which a magnetically polarized molecule with a sufficiently intense magnetic moment can induce a corresponding polarization of valence [and_or other] electrons in the atoms or molecules constituting the walls surface. Once such a polarization is created by induction, magnecules can have rather strong magnetic bonds to said walls.
Magnetically polarized gases are expected to have anomalous absorption or penetration through other substances. This is first due to the reduction of the average molecular volume with inherent increase of permeability, as compared to the same unpolarized gas. The second reason is the magnetic induction of the preceding anomaly.
Magnetically polarized gases are expected to have thermochemical reactions with macroscopic increases of energy releases, as compared to the same reactions among unpolarized gases, an expected anomaly that, alone, has large scientific and industrial significance.
3.4. EXPERIMENTAL EVIDENCE OF MAGNECULES IN GASES
3.4.A. NECESSARY CONDITIONS FOR THE DETECTION OF GAS MAGNECULES
The experimental detection of Santilli's magnecules^TM is difficult due to the fact that conventional analytic equipment and methods have been developed for conventional properties. As such, these instruments are generally ineffective for the measurement of the anomalous properties of
magnecules.
GC-MS are routinely operated at temperatures of the order of 150 C to 250 C, for which no magnecule is expected to exist. To put it explicitly, the conventional operation of GC-MS destroys the very clusters to be detected, and this identifies a first reason for the lack of detection of magnecules until now.
In the absence of IRD, no measurement should be considered to be scientific or final [because of the lack of 50% of the necessary information for a serious conclusion]. Since the great majority of GC-MS are not equipped with IRD, this provides an additional reason why the magnecules have not been detected in until now. In reality, numerous magnecules have been indeed detected in academic laboratories, but were misinterpreted as esoteric molecules precisely because of the lack of IRD.
GC-MS are usually operated with feeding lines with the smallest possible ID, at times of the order of 0.1 mm. This additional unusual requirement is due to Anomaly 7, i.e., the enhanced adhesion of magnecules to the metallic walls of the feeding line, that become clogged-up to the point of preventing the passage of the most interesting magnecules, the big magneclusters with molecular weight in the 1,000's a.m.u. This is another condition analysts with extended practice on conventional gases can only accept after being exposed to the evidence.
This condition identifies another reason why magnecules have simply not been looked for until now. As an illustration, the most interesting molecular weights for Magnegases^TM are those bigger than their heaviest conventional molecule, i.e., bigger than 44 a.m.u. It goes without saying that smaller molecular weights are also important once the analyst seeks an anomaly, rather than a conventional result.
In general, for the evident reason to reduce costs, the ramp time is set at the smallest possible operational value, that is perfectly acceptable for conventional gases, but substantially inappropriate for the anomalies we have to detect. As will be shown, for the case of magnetically polarized gases a small ramp time implies the clustering of all magnecules into one single peak. The analyst then looks at each individual constituent of such a unique large peak, evidently finds fully conventional molecules, thus reaching the "experimental belief" that the peak represents conventional molecules. For a sufficiently large ramp time, the magnecules are instead represented by well separated, large individual peaks. In this case the analyst is forced to identify these peaks individually. Then, and only then, the computer [rather than the analyst] will establish that the MS peaks are "unknown", and confirm the "unknown" character in the absence of IR signature. The latter then establishes the magnecule because it establishes the lack of valence bond for the peak considered.
The first clear experimental evidence of gas magnecules was established by Santilli [8] on June 19, 1998, at the analytic laboratory of NATIONAL TECHNICAL SYSTEMS [NTS] located at McClellan Air Force Base, Sacramento, California. The measurements on were conducted by analysts Louis A. Dee, Branch Manager, and Norman Wade who operated an HP GC model 5890, an HP MS model 5972, and an HP IRD model 5965. Upon inspection on arrival, that the instrument met all conditions 1 to 5, Santilli then, and only then, authorized the measurements.
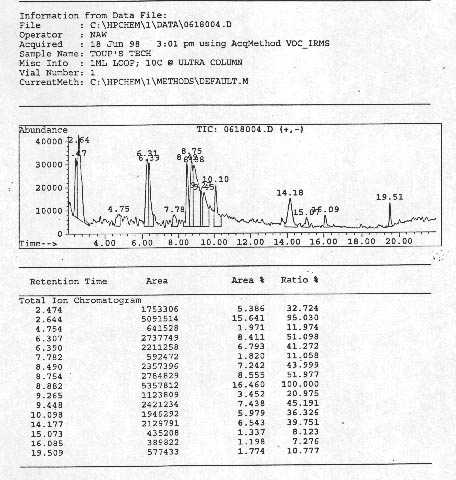
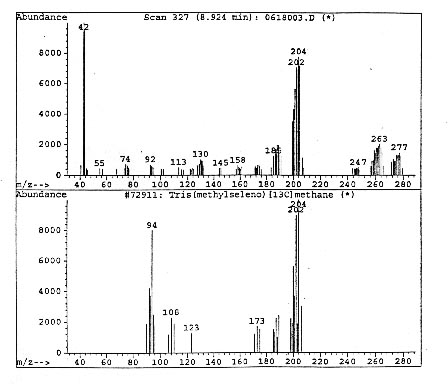
FIGURE 3.10. The anomalous IR signature of CO2 (top) compared to the conventional one (bottom), establishing a polarization of the orbits of valence and other electrons.
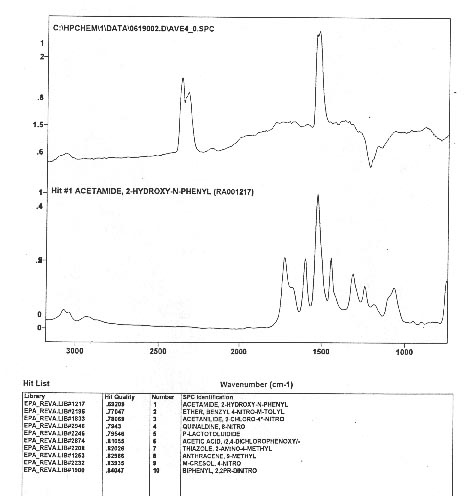
The anomaly of Figure .3.10 is particularly important, inasmuch as it provides experimental evidence for a mutation of the structure of conventional molecules with the presence of additional strong internal binding forces, also expected to be of magnetic type, that is expected to explain the increased energy content.
To appraise the implication of this aspect alone, one should consider the fact that it provides experimental evidence on the polarization of the orbits of internal non-valence electrons, evidently because valence electrons cannot provide any additional molecular bond for both conventional and hadronic chemistry.
The NTS analysts also inspected small peaks in the IR scan, that all resulted to be "unknown", as shown in Figure 3.11, thus completing the experimental evidence of the anomalous IR behavior.
Following the above measurements, the following signed report dated June 23, 1998 was released by the NTS Laboratory [see the original in App. I of Ref. [9]]:
"Dear Dr. Santilli,
Thank you for the opportunity to provide analytic data on AquaFuelTM. Attached are all GC-MS and concomitant GC-IR data that we obtained for the unfiltered sample.
... Our blank runs obtained before and after the analyses show no high mass spectra, thus proving that the high molecular weight species found were actually in the AquaFuelTM sample.
.... Search results using a library of 138,000 chemical compounds, did not indicate any matches with this high molecular weight species found in AquaFuelTM.
Based on the limitations of our equipment and our inability to find a library match, the identification of this mysterious AquaFuelTM ingredient is an unknown.≤
National Technical Systems
Louis A. Dee
Branch Manager"
The measurements on the same sample of magnetically polarized gas tested at NTS were repeated on July 25, 1998, by analyst Kevin Lothridge via a GC-MS_IRD located at the PINELLAS COUNTY FORENSIC LABORATORY [PCFL] of Largo, Florida, under TTL support (for complete documentation see App. II of Ref. [9]).
The equipment consisted of an HP GC model 5890 Series II, an HP MS model 5970 and an HP IRD model 5965B. Even though similar to the equipment used at NTS, the PCFL equipment was significantly different inasmuch as the temperature had to be increased from 10 C to 55 C and the ramp time reduced from 26 minutes to 1 minute. The latter reduction implied the cramping of all peaks of Figure 3.1 into one single large peak, a feature confirmed by all subsequent GC-MS tests with short ramp time.
Despite these differences, the test at PCFL confirmed all Anomalies 1 to 4 first detected at NTS. In addition, the tests provided the first experimental evidence of additional anomalies.
Analyst Kevin Lothridge conducted two MS tests of the same polarized gas sample at different times about 30 minutes apart. As one can see in the original scans [9], the test at PCFL provided the first experimental evidence of Anomaly 5, the mutation in time of magnecules. In fact, the peaks of the same gas at same conditions, but at different times are macroscopically different from each other.
This difference provided evidence that, when colliding, magnecules can break down into ordinary molecules, atoms, and fragments of magnecules, that then recombine with other molecules, atoms, and_or magnecules to form new unknown peaks.
Equivalently, the experimental evidence establishes the expectation that mutation of magnecules can simply occur via the accretion or release of another polarized atoms or molecule without breaking down.
As one can see in the scans [9], the tests conducted by analyst Kevin Lothridge also provided the first experimental evidence that the background following the end of the measurements resulted to be significantly similar to the MS scan, thus providing a direct experimental verification of Anomaly 7 on the anomalous adhesion of magnecules.
The original background prior to the measurements was re-obtained only after flushing the instrument with an inert gas at about 250 C. Such flushing heated the interior walls and parts of the instrument, thus breaking down all magnecules, terminating their anomalous adhesion and re-establishing their conventional molecular structure.
A third independent verification of Santilli's magnecules was conducted on July 29, 1998, by analyst David Fries at the CENTER FOR OCEAN TECHNOLOGY of the UNIVERSITY OF SOUTH FLORIDA [USF] in St. Petersburg, under TTL support. The test were done via the use of a GC-MS Varian 2000 in which the GGC had been removed and replaced by a modified Paul Trap that turned the instrument into a Ion Trap Mass Spectrometer [IT-MS].
Since the equipment belongs to an academic laboratory, it was not equipped with an IRD. This is unfortunate for scientific knowledge, because Paul's Trap is based on a locally strong magnetic field, that is expected to polarize all conventional molecules [although in an amount evidently less than the extreme magnetic fields of an electric arc].
In fact, the background of the instrument prior to the initiation of measurements showed various peaks that were tentatively identified as conventional molecules, from the sole shape in the MS. Only the additional presence and use of an IRD would have permitted the scientific identification of the peaks, because of the sole existence of two possibilities: the IR signatures of the peaks in the background are those of the believed conventional molecules; or said peaks have no IR signature at all, in that case they cannot possibly be conventional molecules.
As one can see in Appendix III of Ref. [9], despite the lack of IRD, the measurements provided a third independent verification of all preceding experimental evidence on the existence of magnecules.
Moreover, the USF measurements provided the first evidence on the existence of magnecules of individual hydrogen atoms. This was due to the accretion or loss by various peaks of only one hydrogen atom due to variations of only one a.m.u. This result was expected because of the known high sensitivity of Paul's Trap. The presence in the magnecules of other individual atoms [such as C or O] is implied in the preceding NTS and PCFL measurements, although it is best established for the case of the hydrogen.
In view of the above occurrence, inspection of the various MS scans indicates the absence in magnecules of systematic numerical periodicities, e.g., their systematic increase with periodicity two [sole accretion of H2 molecules], or thirty-eight [sole accretion of CO molecules].
Experimental evidence on the disappearance of all unknown peaks at sufficiently high temperatures was established for the first time by Dr. N. Palibroda and Dr. P. Glueck at the INSTITUTE FOR ISOTOPIC AND MOLECULAR TECHNOLOGIES [IIMT] of Cluj-Napoka, Romania, also under TTL support, via a GC-MS MAT 311.
When operated at 150 C, the same sample of polarized gas tested at NTS, PCFL, and USF, showed no unknown species at all, and only ordinary light gases with ppm of more complex composites of H, O and C, the latter mostly due to contaminants. This result provided additional support on magnecules as being made-up of ordinary molecules and atoms under a new bond of magnetic origin.
Additional experimental confirmation of the above GC-MS/IRD results have been conducted and are reported in monograph [9].
3.4.C. ORIGIN OF ANOMALOUS ENERGY RELEASE
1) New internal bonds in ordinary molecules. Recall that peaks in the IR scan essentially represent internal molecular bonds. The IR scan of Figure 3.10 is particularly important because it establishes the existence in the CO{sub}2{/sub} molecule of two new internal bonds characterized by the two new peaks. It is evident that these bonds cannot be of valence type, since that bond is already represented by the large IR peak of the same figure. The only possible explanation of the above experimental evidence is therefore that the two new internal bonds are of magnetic type. In turn, the only possible explanation is that the intense magnetic fields of the electric arc polarize also the orbits of internal, non-valence electrons, thus creating new magnetic bonds within the structure of conventional molecules themselves, in addition to magnetic bonds between molecules. Calculations show that these new, internal, magnetic, molecular bonds can store a virtually unlimited amount of energy, thus providing the first explanation of the anomalous energy content of magnegas.
2) Bigger efficiency in chemical reactions. Energies released by chemical reactions in gases, such as the basic reactions
H2 + O2 / 2 -> H2O + 57 Kcal /mol,
have been measured until now for gas molecules in their conventional spherical distributions
of peripheral atomic electrons due to rotations. According to hadronic chemistry [5-7], it is easy to see that, before the above reaction can take place, the orbits of the valence electrons of the hydrogen and oxygen must be polarized in a plane, so as to permit their bonding into the isoelectronium (Figure 3.4). It is evident that magnetically polarized gas molecules admit chemical reactions with an efficiency bigger than that of conventional molecules, because the atoms to be bonded already have the necessary polarization. It then follows that conventional reactions for magnetically polarized gases release more energy than the same reactions for conventional gases, thus providing a second explanation for the anomalous energy content of the class of gases known as Magnegases^TM. Needless to say, the same enhancement of energy release for magnetically polarized gases applies also for all other esoenergetic chemical reactions.
3) Molecular attractions. Ordinary gas molecules have no mutual attractions, again, due to their intrinsic rotations, while magnetically polarized molecules can attract each other, thus implying an additional increase in the efficiency of chemical reactions, such as the synthesis of water. Magnegases have a BTU content of at least 3 times that predicted by quantum chemistry. Their primary chemical reaction during combustion is the burning of hydrogen and oxygen into water. Note that a 3-fold increase of energy release implies the increase of energy released by the water reaction, from 57 Kcal_mole to about 150 Kcal/mole.
In summary, the dramatic increase of energy release by magnetically polarized gases, of at least three times that of unpolarized gases, is the most convincing evidence on the insufficiencies of quantum chemistry in its most important topic, the molecular structure [8].
The anomalous energy content outlined in this section also clarifies the extreme difficulties encountered in the measure of the energy content of magnetically polarized gases. The anomalies establish that all existing calorimeters are not suited for the measurement of energy release under the above anomalies.
Santilli then placed about 50 cc of various samples of perfectly transparent fragrance oils in individual glass containers and exposed them to magnetic fields according to geometries and procedures under patent pending.
Starting with perfectly clear oils of known viscosity, Santilli observed after a few days a visible darkening of the oils, jointly with a visible increase of the viscosity. The changes evidently varied from oil to oil. Both the darkening and the viscosity increased progressively in subsequent days, to reach in certain cases a dark brown color completely opaque to light. The viscosity increased to such an extent that some of the oils lost all their fluidity.
Following the darkening, in certain samples, there was the creation of floating objects that grew in time to such a size to be visible to the naked eye.
These visible effects are of pure magnetic origin because of the lack of any other contribution, e.g., the complete absence of any additives or exposure to any field other than magnetic. After the magnetic treatment, all samples were left undisturbed at ordinary room conditions.
The above tests established beyond scientific doubt that the alteration of the characteristics of the oils was of sole magnetic origin, thus excluding all other possible sources.
Santilli's [loc. cit.] main hypothesis on the reason for the darkness of the oils is that their molecules acquire a magnetic polarization in the orbits of at least some of their electrons [cyclotron resonance orbits], by therefore bonding to each other as it occurs for gases.
3.5.B. PHOTOGRAPHIC EVIDENCE OF MAGNECULES IN LIQUIDS
Figures 3.12 refers to the GR fragrance oil received under the code "ING258AIN, Test 2" subjected to the rudimentary magnetic polarization indicated in the preceding section under the respective magnification 10X and 100X.
As one can see, these photographs establish that, under the indicated magnetic treatment, the oil has acquired a structure of the type of "brick layering" that is visible under only 10X magnification [top of Figs. 3.12]. This layering is, per se, highly anomalous for a liquid that was originally fully transparent. Note that Santilli's magnecules are not constituted by the individual "bricks" shown in the 100X magnification [bottom of the figures], but rather by the dark substance thjat interlock said "bricks".
Inspection of the various photographs shows a variety of sizes of magnecules, thus establishing their lack of unique characteristics for any given oil. This evidently confirms the lack of a valence bond [because the latter would imply a fixed maximal size]. The photographs also show the magnecules capability for accretion, that is, the capability of increasing their size via the addition of further oil molecules.
FIGURE 3.12: First photographic evidence of Prof. Santilli's new chemical species of
magnecules in liquids obtained on January 28, 1999, by Dr. Konrad Lerch,
Director of the Givaudan-Roure Research Laboratory in Dubendorf,
Switzerland, on the GR fragrance oil "ING258IN Test 2" under
magnification 100X. The new chemical species of Magnecules was instrumental for the subsequent discovery by Prof. Santilli of the over-unity PlasmaArcFlow Reactor and its applications.
3.5.C SPECTROSCOPIC EVIDENCE OF MAGNECULES IN LIQUIDS
The measurements were done on: Sample 1, pure [magnetically untreated] GR "Fragrance Oil 2"; Sample 2, magnetically untreated tap water; and Sample 3, a magnetically treated mixture of the two.
It should be noted that the Tekmar equipment lacks the computer search as well as the UV scan. Also, recall that magnecules in a light gas can have molecular weight all the way to 10,000 a.m.u. and more.
.
Comprehensive tests via a very modern equipment for Liquid Chromatographic Mass Spectroscopy [LC-MS] with UltraViolet Detector [UVD] were conducted on the same GR fragrance oil "ING258IN Test 2" of Figures 3.12, 3.13, and 3.13, on December 1, 1998, by Prof. Kenneth G. Furton, Chairman, and Prof. Piero R. Gardinali, Laboratory Director, DEPARTMENT OF CHEMISTRY of FLORIDA INTERNATIONAL UNIVERSITY [FIU], Miami, Florida [8]. The tests were then repeated on December 17 and 18 by confirming the preceding results.
The tests were conducted under a number of technical characterizations specifically selected to detect magnecules, among which:
1) Total Ion Chromatogram [TIC] collected under the positive ion atmospheric pressure electrospray ionization [ESI+] mode;
The tests were conducted on the following samples:
A) Sample GR331, the magnetically untreated, fully transparent GR fragrance oil "ING258IN Test 2";
B) Sample GR332, magnetically treated ING258IN Test 2 with 10 % DiproPylene Glycol [DPG];
C) Sample GR332S, bottom layer of the preceding sample;
D) Sample GR335, magnetically treated mixture 4 % GR fragrance oil ≥ING258IN Test 2≤, 0.4 % DPG and 95 % tap water;
E) Sample GR335O, visible dark clusters in the preceding sample.
These tests provided conclusive experimental evidence on the existence of Santilli's magnecules in liquids. To avoid a prohibitive length of this presentation, in the following we reproduce only a few print-out of the comprehensive and detailed documentation obtained at FIU [that is available in its entirety as Appendix V of Ref. [9].
FIGURE 3.15: FIU scan on the untreated GR oil "ING258IN Test 2" of Figures 3.12, 3.13, and 3.14.
3.5.D. MAGNETIC MUTATION OF
PHYSICAL CHARACTERISTICS IN LIQUIDS
The reader should be aware that we are referring to large macroscopic alterations often visible to the naked eye, that are at times such to alter completely the original features, as it was the case the complete loss of transparency and fluidity in the magnetically treated oils of the preceding sections.
At a deeper level, it appears that the mutation of physical characteristics are due to various contributions, such as:
1) the elimination under a suitable magnetic field of [at least some of the] rotational motions of the molecules that, per se, implies a decrease of their average volume with consequential increase of the specific density;
2) the polarization of the orbits of the valence electrons that imply an additional decrease of the average molecular volume and consequential increase of the specific density, viscosity and other characteristics; and
3) the magnetic bond among different molecules that implies additional contributions to the indicated mutations.
In this section we report direct experimental evidence on measurements of specific density, viscosity and other characteristics first presented in Ref. [8], that confirms the above prediction.
The samples used ordinary tap water and a number of GR fragrant oils. All samples here considered were prepared by conventionally mixing tap water and one fragrant oil and then submitting that mixture to rather weak permanent magnets of 200 G [much weaker than those used for Pictures 3,12] via an equipment under patent pending. All samples resulted to be very stable at ordinary conditions without any measurable change over a period of about year.
In early 1998, Santilli [loc. cit.] prepared various samples along the above procedure numbered from 1 to 25. The measurements of the specific density were conducted on March 9, 1998 by analyst Dr. A. Sibille at the SGS U.S. TESTING COMPANY, INC. [USTC] of Fairfield, New Jersey. The results of the tests are presented in derails in Refs. [8.9].
In the transition from Sample 1 [untreated water] to Sample 2 [magnetically treated water] there is an increase of the specific density in the macroscopic amount of 0.86% thus confirming the indicated magnetic mutation of water. In turn, the increase in density supports the existence of magneplexes in magnetically treated water.
As well known, fragrance oils are [generally] lighter than water, i.e., the specific density of the untreated fragrance is smaller than that of the untreated water. According to quantum chemistry, the specific density of any mixture of the above two liquids, whether solution, suspension or dispersion, must be in between the lighter and heavier specific densities.
On the contrary, the specific density of the magnetically treated mixture of GR ≥APC fragrance 1≤ with tap water, resulted to be bigger than that of the densest liquid, the water. This measurement constitutes additional, rather strong, direct experimental verification of the magnetic mutation of physical characteristics in liquids.
The reader should be made aware that the above anomaly [a mixture of two liquids whose specific density is bigger than that of the heaviest of the two] cannot be explained via conventional quantum chemical knowledge.
` On the contrary, the anomaly is theoretically predicted and quantitatively treatable by the covering hadronic chemistry [5-7] via the creation of magnecules.
A remarkable feature is that the magnetic mutations of density are macroscopically large. In fact, they were called by an analyst "UPS-type anomalies", meaning that the shipment via UPS of a given volume of a magnetically treated liquid requires an increase of the shipping cost due to the macroscopic increase of the weight.
A further prediction of magnetically polarized liquids is the increase of its viscosity . This is evidently due to the arbitrary size of an individual magnecule, as well as the tendency of the same to bond near-by molecules, resulting in accretions, not to mention the anomalous adhesion to the walls of the container that has been systematically detected for all magnetically polarized liquids.
As indicated earlier, in certain cases the increase of viscosity is so large to be first visible to the naked eye and, when the treatment is sufficiently protracted, the increase of viscosity is such to lose the customary liquid mobility.
The measurements on viscosity are reported in Refs. [8,9]. They were done on March 9, 1998 by analyst J. R. Tyminski at SGS U.S. TESTING COMPANY, INC. of Fairfield, New Jersey. The selected sample was an engine oil 30-40 Castrol Motor Oil subjected to magnetic treatments via two different equipment called of Type A and B. All treatments were done at ordinary conditions in the outsider of the oil container without any additive or change of conditions of any type. Yet the measurements established an increase of viscosity of 51.2%.
The tests on viscosity also provide evidence of the anomalous adhesion of liquids with magnecules that is established in this case by a dramatic, macroscopic increase of adhesion of the oil to the walls of the glass container, an adhesion that is visible by the naked eye.
The same macroscopic anomaly is confirmed at the microscopic level. During the measurement of viscosity there was such an anomalous adhesion of the magnetically treated oils to the walls of the instrument that could not be removed via routine cleaning with acetone and required the use of strong acids.
This anomalous adhesion is further experimental evidence on the existence of Santilli's magnecules because of their predicted capability to induce the polarization of the orbits of the valence electrons of the atoms in the walls of the container, thus resulting in anomalous adhesion via magnetic bonds due to induction.
It is evident that the magnetic mutations of density and viscosity implies the expected mutation of all other physical characteristics of the liquid considered. Measurements along these lines are solicited.
The existence of mutation of physical characteristics then implies the mutation of chemical features. An illustration is given by the visual evidence reported by the analysts of USTC according to which the reaction of magnetically treated oils with acetone is dramatically different than that with untreated oil, beginning with a visual mutation in color, texture and other appearances.
3.5.E. EVIDENCE OF SANTILLI'S MAGNECULES IN SOLIDS
Needless to say, the above simplest possible evidence is only the beginning of various possibilities, such as the true chemical composition of solids created under intense magnetic fields, such as fullerenes {5} and other composites.
These studies are under way and their outcome will be uploaded in this web site when available.
An additional experimental verification has been recently conducted by analyst Dr.
Jeffrey Riggs, director of the National technical Laboratories in North Highlands, California, that have confirmed ALL predictions by Prof. Santilli in his original memoir [8], as well as all preceding analyses.
The analyses were conducted, specifically, with Santilli's Magnegas^TM produced by
TTL Recyclers (see the preceding Part 2 and the web site http://www.toupstech.com.
A copy of the rather voluminous documentation is available in the forthcoming monograph [9]. For brevity we here reproduce below the official letter by the analyst, as well as two representative scans.
January 24, 2000
The analysis of the Magnegas^TM air samples gave no conclusive mass spectra data. The computer library of 138 compounds was searched for matching spectra. No matches were obtained by the instrument, either in the low or in the high temperature methods. There were large peaks that would appear randomly in the analyses, but the computer was unable to identify any of the peaks that were seen.
The anomalies of the large peaks that were detected in the GCMS traces even remained in the blanks that were analyzed after the samples. The interpretation of these peaks is beyond the scope of the instrumentation used. Therefore, the identity of these peaks remains a mystery.
The FTIR was able to identify carbon monoxide, carbon dioxide, and the presence of hydrocarbons due to the carbon-hydrogen stretches at the 3000 cm^-1 range.
Since the FTIR in the GCMS could not be used, no real time trace of any corresponding peaks on the mass spectra output could be compared to the FTIR. However, as per conversation with Dr. santilli, an FTIR with a long path gas cell (approximately 20 meters) was used instead. It gave evidence of carbon monoxide, carbon dioxide, and hydrocarbons due to the various wavelengths seen. Other infrared spectra were detected also, but the spectra were beyond the capabilities of the instrument used to identify the samples any further.
All of the GCMS and FTIR scans and library searches are included in the data package. Also, with the FTIR data is included the reference spectra of several of the compounds observed.
Analyst:
As the visitor can see, the NTS analyses identified the existence of peaks in the GCMS none of that could be identified and that have no IR counterpart. Similarly, the IR detector fully identified CO, CO2, and other conventional molecules that had no identification in the GCMS. This evidence establishes that the peaks do not possess a valence structure, and all the conventional molecules identified in the FTIR are necessary constituents of the unidentified peaks in the GCMS.
In his letter, Dr. Riggs explicitly confirm the anomalous adhesion of magnecules^TM
as predicted by Santilli [8], since the blanks after the tests show essentially the same spectra as the original gas. Since electrostatic bonds are known to be very weak and unstable, this establishes that the anomalous adhesion by Magnegas to the instrument is of magnetic origin. By recalling that the gas samples were created under very intense magnetic fields, these data establish that the unidentified peaks in the GCMS are Santilli's magnecules^TM.
Close inspection of the rather voluminous documentation provides experimental verifications of each and every one of the anomalies of ,magnecules predicted by Santilli. For brevity we refer to monograph [9], and to the example of Figures 3.17 and 3.18 below.
************************************
REFERENCES
[1] Ruggero Maria Santilli
[2] Ruggero Maria Santilli
[3] A. O. E. Animalu, Hadronic J. Vol. 17,p. 349 (1994).
[4] A. O. E. Animalu and R. M. Santilli, Intern. J. Quantum Chemistry
Vol. 29, p. 175 (1995).
[5] R. M. Santilli and D. D. Shillady, A new isochemical model of the hydrogen molecule, Intern. J. Hydrogen Energy, Vol. 24, p. 943 (1999).
[6] R. M. Santilli and D. D. Shillady, A new isochemical model of the water molecules, Intern. J. Hydrogen Energy, Vol. 25, p. 173 (2000).
[7] R. M. Santilli and D. D. Shillady, Ab Initio hadronic chemistry, Hadronic Press,
in press science2@gte.net
[8] R. M. Santilli, Hadronic J. Vol. 21, p. 789 (1998).
[9] R. M. Santilli, The New Chemical Species of Magnecules: Its applications and Experimental verifications, Hadronic Press,
in press science2@gte.net
[10] M. G. Kucherenkko and A. K. Aringazin, Hadronmic J. Vol. 21, p. 895 (1998).
[11] H. Eldridge, U. S. Patent no. 603,058, 1898 [11a]. W. A. Dammann and D. Wallman, U. S. Patents nos. 5,159,900 (1992) and 5,417,817 (1995) [11b]. W. H. Richardson, jr., U. S. Patents nos. 5,435,274 (1995), 5,692,459 (1997), 5,792,325 (1999) [11c].
APPENDIX: TABLE OF CONTENTS OF SANTILLI'S REF. [1,2], "THE PHYSICS OF NEW CLEAN ENERGIES AND FUELS"
PART II: THE NEW MECHANICS, 46
PART III: NEUTRON STRUCTURE AND NEW ENERGIES
OF CLASS I, 84
PART IV: STRUCTURE OF NUCLEI AND NEW ENERGIES OF CLASS II, 143
PART V: STRUCTURE OF MOLECULES, NEW CLEAN FUELS, AND
Main Page
| Table of Contents | Journals
| Subscriptions | Submissions
| Monographs | IBR
Grants | IBR Conferences | Hadronic
Mechanics | Latest Discoveries | About
Us | CV of IBR Members | E-Mail
Us
Copyright ©
1997-2002 Institute for Basic Research, P. O. Box 1577, Palm Harbor, FL 34682, U.S.A.
As indicated earlier, a most significant aspect of the class of magnetically polarized gases is their anomalous energy content. According to quantum chemistry,
3.5. EXPERIMENTAL EVIDENCE OF SANTILLI'S MAGNECULES IN LIQUIDS
3.5.A. FORMATION OF MAGNECULES IN LIQUIDS
The way in which Santilli [8] first created magnecules in liquids is the following. In early 1998 he obtained a number of samples of fragrance oils thanks to the courtesy of the multinational giant in the fragrance industry, GIVAUDAN ROURE CORPORATION
[GR].
The alteration of the structure of fragrance oils under suitable magnetic exposures was first confirmed by photographs taken with a microscope with minimal magnification, as illustrated in the pictures of Figures 3.12 (see monograph [9] for several other photographic evidence).

The first experimental evidence of Santilli's magneclusters in liquids was established on May 5, 1998, by analysts Brian Wallace and Mia Burnett at TEKMAR-DOHRMANN CORPORATION [TDC] in Cincinnati, Ohio, operating a Tekmar 7000 HT Static Headspacer Autosampler equipped with a Flame Ionization Detector [FID]. The tests were repeated on May 8 and 11, 1998, by confirming the preceding results.
FIGURE 3.13. A reproduction of one of the scans conducted on May 5, 1998, at 8.18 a.m., by analysts Brian Wallace and Mia Burnett at TEKMAR-DOHRMANN CORPORATION [TDC] in Cincinnati, Ohio, by operating a Tekmar 7000 HT Static Headspacer Autosampler equipped with a Flame Ionization Detector [FID]. The scan is for exactly the same sample of GR fragrance oil of Figure 3.12, although for its magnetically unpolarized form. The oil results to be composed by three primary molecular constituents with: Peak 1 at 6.430 min and 24.28 %; Peak 2 at 8.604 min and 3.19 %; and Peak 3 at 37.742 and 70.00 % [the latter one being off scan scale], totaling 97.57 %.
FIGURE 3.14. A reproduction of some of the various scans conducted by TDC on the magnetically polarized version of the GR fragrance oil of the scan of Figure 3.13. Santilli's magnecules are established by the appearance of the "unknown" peaks in large percentages that do not exist in the magnetically untreated oil. Note numerous additional variations, such as the decrease of the percentage of the conventional molecular peaks (see monograph [9] for the complete reproduction of all peaks, including the related percentages).
2) Integrated TIC with retention times and areas for the most abundant peaks;
3) Raw mass spectra for all peaks identified in item 2;
4) HPLC chromatograms collected at fixed wavelength of 254 cm;
5) UV-visible spectra form the HPLC diode array detector from 230- to 700 mm.
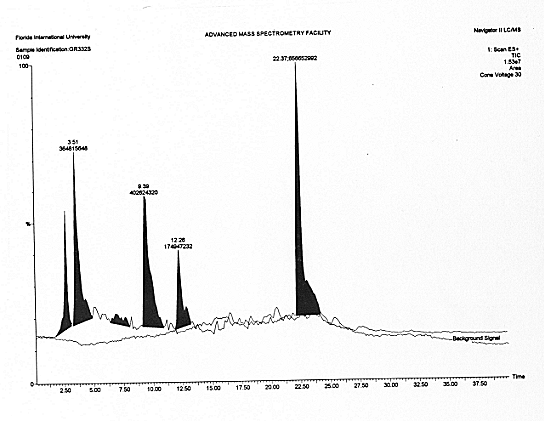
FIGURE 3.16: FIU scan on the chemical structure of the magnetically treated GR oil "ING258IN Test 2". Santilli's magnecules in liquids is established by the large new peak on the r.h.s.
The existence of SANTILLI'S magnecules in liquids implies necessary alterations, called mutations [8], of physical characteristics, such as increase of the specific density and viscosity. This is due to the fact that magnetic bonds among ordinary molecules imply an evident reduction of intermolecular distances, thus resulting in more molecules per unit volume as compared to the magnetically untreated liquid. The increase in viscosity is then consequential.
The direct experimental evidence on the existence of magnecules in solids available at this writing is given by the evidence that all liquids with magnecules preserve the new species when frozen, as established by the recovering of anomalous features when returning to the liquid state, including the recovering at the liquid state of opacity and other features prior to freezing.
3.5.F. ADDITIONAL EXPERIMENTAL EVIDENCE OF SANTILLI'S MAGNECULES^TM
prepared by J. V. Kadeisvili
As indicated earlier in this web page, Santilli's magnecules were first established experimentally in June 1998 at the Mc\Clellan Air Force Base near Sacramento, California, and then confirmed in July 1998 the subsequent month of July at the Pinellas Forensic Laboratory, Florida, in November 1998 at the university of South Florida in St Petersburg, Florida, and in January 1999 at the analytic laboratory of the Chemistry Department of Florida International University in Miami.
FROM: National Technical Systems, North Highland, CA
TO: Toups Technology Licensing, Largo, Florida
SUBJECT: Analysis of Santilli's Magnegas^TM via GCMS and FTIR
Jeffrey Riggs
FIGURE 3.17: A reproduction of one of the numerous scans conducted on Santilli's
Magnegas^TM in mid January 2000 by Dr. Jeffrey Riggs of NTS Analytic Laboratories in North Highland, California, that establishes the unknown nature of the chemical composition of the gas, following a computer search among 138,000 molecules.
FIGURE 3.18: A reproduction of another GCMS scan at NTS conducted on exactly the same gas, on the same GCMS equipment, under the same conditions (except for a longer ramp time), that establishes the mutation of Santilli's
magnecules. In fact, as one can see, the clusters of this scan are different than those of the preceding scan. Note also in the same cluster the variation from 97 amu to 98
amu, that can only be explained via the accretion of one individual hydrogen atom. This confirms the presence in Santilli's magnecules of individual atoms, besides ordinary molecules. The difference between 55 amu and 57 amu then establishes the accretion of the magnecule by one hydrogen molecule. Other Santilli mutations have a variety of interpretations.
as of early 1999
THE PHYSICS OF NEW CLEAN ENERGIES AND FUELS
AS PREDICTED BY HADRONIC MECHANICS,
Papers I, II, III, IV, and V
JOURNAL OF NEW ENERGIES, Vol. 4, issue no. 1, 1999
"THE PHYSICS OF NEW, CLEAN, ENERGIES AND FUELS",
as Predicted by Hadronic Mechanics"
Ukraine Academy of Sciences, Kiev,
Distributed in the USA by
Hadronic Press
science2@gte.net
PART I: INSUFFICIENCIES OF QUANTUM MECHANICS, page 10
1. INTRODUCTION, page 10
1.1. The Fundamental Open Problem of the Emerging New Energies, 10
1.2. Definition of New Energies, 13
1.3. Statement of Objectives, 15
2. INSUFFICIENCIES OF QUANTUM MECHANICS IN VARIOUS FIELDS, 16
2.1. Conditions of Validity of a Given Theory, 17
2.2. Insufficiencies of Quantum Mechanics in Atomic Physics, 19
2.3. Insufficiencies of Quantum Mechanics in Nuclear Physics, 21
2.4. Insufficiencies of Quantum Mechanics in Particle Physics, 29
2.5. Insufficiencies of Quantum Mechanics in Superconductivity, 32
2.6. Insufficiencies of Quantum Mechanics in Gravitation,
Astrophysics and Cosmology, 33
2.7. Insufficiencies of Quantum Mechanics in Chemistry, 33
2.8. Insufficiencies of Quantum Mechanics in Biology, 36
3. INSUFFICIENCIES OF SPECIAL AND GENERAL RELATIVITIES, 38
3.1. Statement of Objectives, 38
3.2. Insufficiencies of Special Relativity, 38
3.3. Inconsistencies of General Relativity, 40
1.INTRODUCTION, 46
2.ORIGINS OF THE INSUFFICIENCIES OF QUANTUM MECHANICS, 49
3.TE ≥MAJESTIC≤ AXIOMATIC CONSISTENCY OF QUANTUM MECHANICS, 50
4.CATASTROPHIC INCONSISTENCY OF BROADER THEORIES BASED ON
CONVENTIONAL MATHEMATICS, 52
5. HE NOVEL ISO≠, GENO≠ AND HYPER≠MATHEMATICS AND THEIR
ISODUALS, 56
6.CLASSICAL AND OPERATOR ISO-, GENO-, AND HYPER-MECHANICS, 61
6.1. Introduction, 61
6.2. Nonrelativistic Classical Isomechanics, 62
6.3. Nonrelativistic Isoquantization, 63
6.4. Nonrelativistic Isomechanics, 64
6.5. Relativistic Isomechanics, 71
6.6. Geno- and Hyper-mechanics, 71
7.ISO-, GENO-, HYPER-RELATIVITIES AND COSMOLOGIES, 74
8.ISO-, GENO-, AND HYPER-CONDUCTIVITY, 79
9.ISO-, GENO- AND HYPER-CHEMISTRY, 80
10.SIMPLE CONSTRUCTION OF CLASSICAL AND OPERATOR GENERALIZED
THEORIES, 80
1.CLASSIFICATION OF NEW ENERGIES, 84
2.EXPERIMENTAL VERIFICATIONS OF HADRONIC MECHANICS IN
PARTICLE PHYSICS, ASTROPHYSICS, AND COSMOLOGY, 87
2.1. Statement of Objectives, 87
2.2. Insufficiencies of Special Relativity and the
Universality of its Isotopic Covering, 87
2.3. Experimental Verification with Data on the Local Speed of Light, 88
2.4. Experimental Verifications Via Phenomenological
Calculations of Hadronic Media, 90
2.5. Experimental Verification with the Behavior of
the Meanlife of Unstable Hadrons with Speed, 91
2.6. Experimental Verification Via Data on the
Bose-Einstein Correlation, 95
2.7. Experimental Verifications in Gravitation, 99
2.8. Experimental Verifications in Astrophysics, 101
2.9. Experimental Verifications in Cosmology, 104
3. THE NEW STRUCTURE MODEL OF HADRONS WITH PHYSICAL CONSTITUENTS, 106
3.1. Statement of Objectives, 106
3.2. The Forgotten Legacy of Rutherford, 107
3.3. Hadronic Two-Body Bound States, 109
3.4. The Structure Model ľō = (e+, e-)HM, 114
3.5. The Structure Model n = [pĢ+, eĢ-]HM, 117
3.6. Don BorghiĻs Verifications of the Synthesis of
the Neutron from Protons and Electrons, 123
3.7. Compatibility of the New Structure Model of
Hadrons with SU(3)-Color Classification, 125
4. APPLICATION TO NEW ENERGIES OF CLASS I, 127
4.1. The Stimulated Decay of the Neutron, 127
4.2. Hadronic Energy (Copyrighted and Patent Pending), 129
4.3. TsagasĻ Experimental Verification of Hadronic Energy, 133
4.4. Application to the Stimulated Decay of Nuclear Waste
(Copyrighted and Patent Pending), 135
4.5. Physical Laws of New Energies of Class I
(Copyrighted and Patent Pending), 137
5.PROPOSED NEW EXPERIMENTS FOR NEW ENERGIES, 138
1. INTRODUCTION, 143
1.1. Statement of Objectives, 143
1.2. Lack of Exact Character of Quantum Mechanics in Nuclear Physics, 146
1.2.1.Insufficiencies of quantum mechanics for the nuclear force, 146
1.2.2.Insufficiencies of quantum mechanics for the nuclear structure, 147
1.2.3.Insufficiencies of quantum mechanics for the representation
of nuclear magnetic moments, 148
1.2.4.Insufficiencies of quantum mechanics for the
representation of other nuclear data, 149
1.2.5.Insufficiencies of quantum mechanics for the representation
of dissipative nuclear reactions, 150
1.3. Primary Reasons for the Insufficiencies of Quantum
Mechanics and the Selection of a Covering Theory, 150
2. APPLICATIONS AND VERIFICATIONS OF HADRONIC MECHANICS
IN NUCLEAR PHYSICS, 153
2.1. Main Structural Lines, 153
2.2. Reconstruction of the Exact Isospin Symmetry, 155
2.3. Exact Representation of Total Nuclear Magnetic Moments, 158
2.3.1.The historical hypothesis, 159
Incompatibility of the historical hypothesis with the special relativity, 159
2.3.3.Compatibility of the historical hypothesis with
the isospecial relativity, 160
The isodirac equation, 161
2.3.5.Conventional values of angular momentum and spin,
yet mutated magnetic moments, 162
2.3.6.Exact representation of the deuteron magnetic moment, 163
2.3.7.Exact representation of all remaining nuclear magnetic moments, 165
2.4. Exact Representation of RauchĻs 4ľ Interferometric Measurements, 165
2.5. Hadronic Model of the Deuteron Structure, 171
2.5.1.Conceptual foundations for nuclei, 171
2.5.2.Conceptual foundations for the deuteron, 171
2.5.3.Deuteron stability, 173
2.5.4.Deuteron size, 173
2.5.5.Deuteron charge, 174
2.5.6.Deuteron spin, 175
2.5.7.Deuteron force, 176
2.5.8.Deuteron binding energy, 178
2.5.9.Deuteron total energy, 178
2.5.10.Deuteron magnetic moment, 180
2.5.11.Deuteron electric dipole moment and parity, 181
2.6.Nuclear Forces, 181
2.7. Nuclear Models, 182
3. PHYSICAL LAWS OF NEW ENERGIES OF CLASS II AS PREDICTED BY
HADRONIC MECHANICS, 182
3.1.Basic Equations for stimulated Nuclear Transmutations, 182
3.2.Physical Laws of New Energies of Class II (Copyrighted
and Patent Pending), 188
3.3.The Hadronic Lithium Reactor (Copyrighted and Patent Pending), 190
3.4.The Hadronic Helium Reactor (Copyrighted, Patent Pending), 195
3.5.The Hadronic Nitrogen Reactor (Copyrighted and Patent Pending), 198
3.6.Hadronic Reactor to Synthesize Natural Element (Copyrighted
and Patent Pending), 202
3.7. Concluding remarks, 202
NEW CLEAN, ENERGIES OF CLASS III, 205
1. INTRODUCTION, 206
1.1. Statement of Objectives, 206
1.2. Major Environmental Problems Addressed
by Toups Technology Licensing, 207
1.4. Hadronic Mechanics, Superconductivity, and
Chemistry, and the New Model of Electron Bonding, 215
1.5. The New Chemical Species of Magnecules, 222
2.EXPERIMENTAL VERIFICATION OF HADRONIC MECHANICS IN
SUPERCONDUCTIVITY AND CHEMISTRY, 228
2.1. Conceptual Foundations of Electron Bonding and Clustering, 228
2.2. Basic Equations for Electron Bonding and Clustering, 231
2.3. Experimental Verifications in Superconductivity, 235
2.4. Experimental Verifications in the Hydrogen Molecule, 239
2.5. Experimental Verification in the Water Molecule, 245
3.NEW COMBUSTIBLE GASES RESOLVING THE ALARMING ENVIRONMENTAL PROBLEMS
CAUSED BY FOSSIL FUELS DEVELOPED AT TOUPS TECHNOLOGY LICENSING, 249
3.1. Creation of Magnecules in Gases, 249
3.2. Definition of Magnecules and their Anomalies
(Copyrighted and Patent Pending), 251
3.3. Necessary Conditions for the Detection of Magnecules
(Copyrighted and Patent Pending), 253
3.4. Spectroscopic Evidence of Magnecules in Gases.
(Copyrighted and Patent Pending), 255
3.5. Origin of Anomalous Energy Content
(Copyrighted and Patent Pending), 269
3.6. Creation of Magnecules in Liquids
(Copyrighted and Patent Pending), 270
3.7. Photographic Evidence of Magnecules in Liquids
(Copyrighted and Patent Pending), 271
3.8. Spectroscopic Evidence of Magnecules in Liquids
(Copyrighted and Patent Pending), 271
3.9. Experimental Verification of Mutated Physical
Characteristics (Copyrighted and Patent Pending) , 274
3.1 Evidence of magnecules in solids (Copyrighted and Patent Pending), 282
3.11.Industrial Applications of the New TTL Technology of Magnetically Polarized Substances (Copyrighted and Patent Pending), 282
3.12.The New TTL MagneGasTM Resolving the Alarming Environmental
Problems Caused by Fossil Fuel (Copyright, TradeMark,
and Patent Pending), 283
4.NEW CLEAN ENERGIES OF CLASS III, 287
4.1. Introduction, 287
4.2. Physical Laws of New Energies of Class III
(Copyrighted and Patent Pending), 288
4.3. Application of Hadronic Mechanics to the
Electromagnetically Pinched, 289
Deuterium Energy (Copyrighted and Patent Pending), 289
4.4. Application of Hadronic Mechanics to Cold Fusion
(Copyrighted and Patent Pending), 292
4.5. Application of Hadronic Mechanics to MillsĻ Blacklight
(Copyrighted and Patent Pending), 304
5.CONCLUDING REMARKS, 307
ACKNOWLEDGMENTS, 308
APPENDIX 1: THE PLASMA-ARC-FLOW HADRONIC REACTOR AND ITS OVERUNITY
BIBLIOGRAPHY:br>
INDEX
Tel: 1-727-934 9593 Fax: 1-727-934 9275 E-Mail: ibr@gte.net
All Rights Reserved.